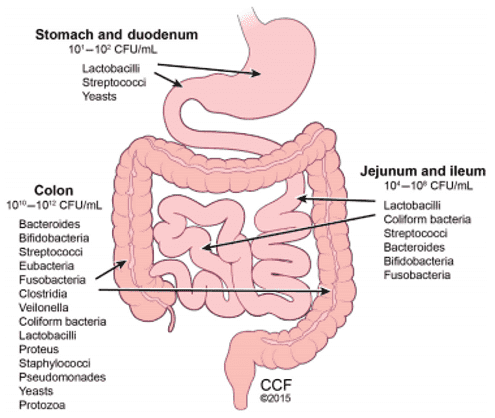
Our gastrointestinal tract is home to a large diversity of microorganisms, which play significant roles to human health. By profiling the composition and alteration of gut microbiota, the research based on next-generation sequencing technologies (targeted sequencing and metagenomic sequencing) has highlighted the structure and physiological function of the gut microbiota. Since there is a body of knowledge with established clues as to the effect of the gut microbiome on human health, there have been ideas conceived for a more personalized approach to healthcare. An example that is already in practice to address some symptoms of diseases is ‘fecal transplantation', or transfer of stool from a healthy donor to a patient plagued with Clostridium difficile colitis which causes diarrhea and abdominal cramps. Currently, it is being used as an alternative if the patient develops complications due to antibiotic therapy, but would need stool DNA tests of the specific patient. The procedure is still considered experimental. While another more common method being practiced is the dietary intervention.
On the other hand, depending on the route of circulation the ingested material will undergo, the gut microbiome can interfere with the uptake of the drug ingested as it breaks down the drug further and enables faster excretion before it can effectively reach its target organ. Digoxin, for example, is observed to be excreted in some patients who are being treated for irregular heartbeat and heart failure. Ingested drugs might also incur changes in their chemical structure that would affect their effectiveness as a treatment to the patient. Toxicity or half-life, the time required for the drug to be decreased to half its concentration in the body, may increase causing lingering side-effects. This is due to the fact that enzymes produced by the microbiome compete with human enzymes involved in drug detoxification, leading the toxic metabolites of the drug ingested for therapeutic purposes to build up, thus, prolonged negative effects on the human constitution. An example of this would be the use of drugs used in cancer treatments.
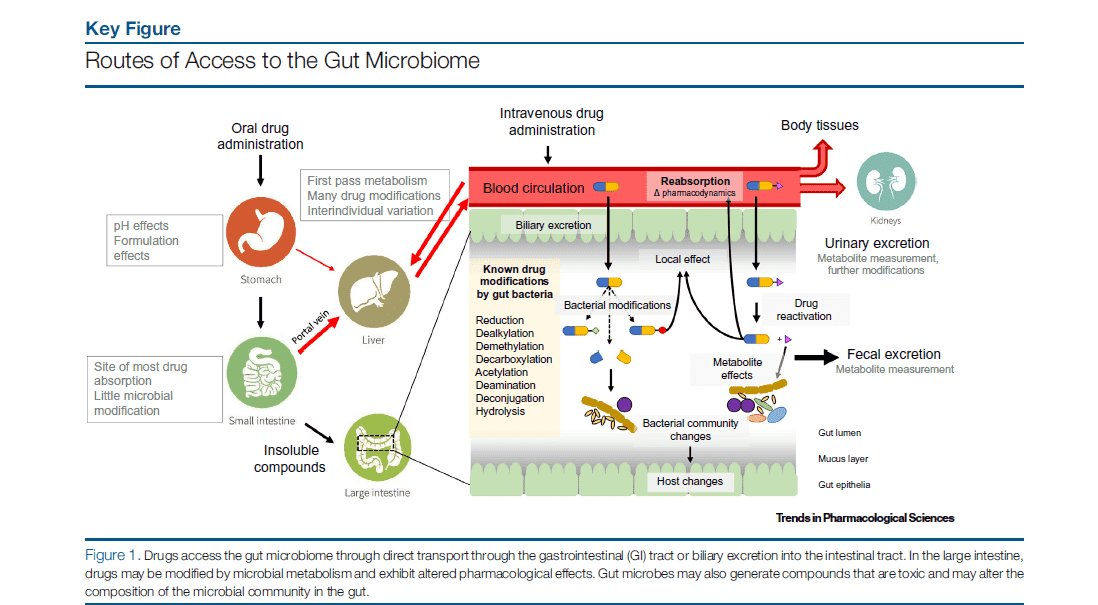 Figure 1. The effects of the gut microbiome on drug intake. (Hitchings, 2019)
Figure 1. The effects of the gut microbiome on drug intake. (Hitchings, 2019)
Moving forward though, further research on the gut microbiome in human health could innovate drug development so that the chemical structure of the drug being taken may be improved for more efficient delivery and metabolism of the patient. The use of biomarkers due to the differences in gut microbiome composition among individuals may solidify the relationship of disease, and diet needed by the patient. Biomarkers may also be used to learn the exact dosage of drugs to be used for optimal effect in conjunction with the patient's microbiome. Drug therapy may also be further improved by genetic screening of the patient's gut microbiome, to learn of the possible activity of drugs to be ingested or, may allow interventions targeting microbial enzymes that target human enzymes used in the detoxification process.
All in all, there is a multitude of possibilities to address disease prevention, development, and treatment for a higher quality of life, even for predisposed individuals. These studies will then help further advance 'precision medicine', and create a personalized pharmacy to suit our individual needs.
References
- Hitchings R, Kelly L. Predicting and understanding the human microbiome's impact on pharmacology. Trends in pharmacological sciences. 2019, 40(7):495-505.
- Spanogiannopoulos P, Bess EN, Carmody RN, Turnbaugh PJ. The microbial pharmacists within us: a metagenomic view of xenobiotic metabolism. Nature Reviews Microbiology. 2016, 14(5):273.