Functional Analysis
Figuring out what the organisms in your specimen is a major feature of metagenomic analysis. There are many methods to do this, but querying a particular set of known patterns that have a related function or can be linked to a series of processes is one common and practical approach. Examples of methods that are different are:
Gene Prediction: It almost always involves a formed sequence that is not guaranteed by metagenomic datasets, although this is often a need of novel genes/organisms.
Associated Functional Inference of Marker Gene: Very helpful for targeted metagenomics. If an associated gene is evident, it can deduce functions.
Marker Gene Analysis: the fastest/most straightforward approach- Look your readings against a database of known functional genes.
Microbial Sequencing Methods
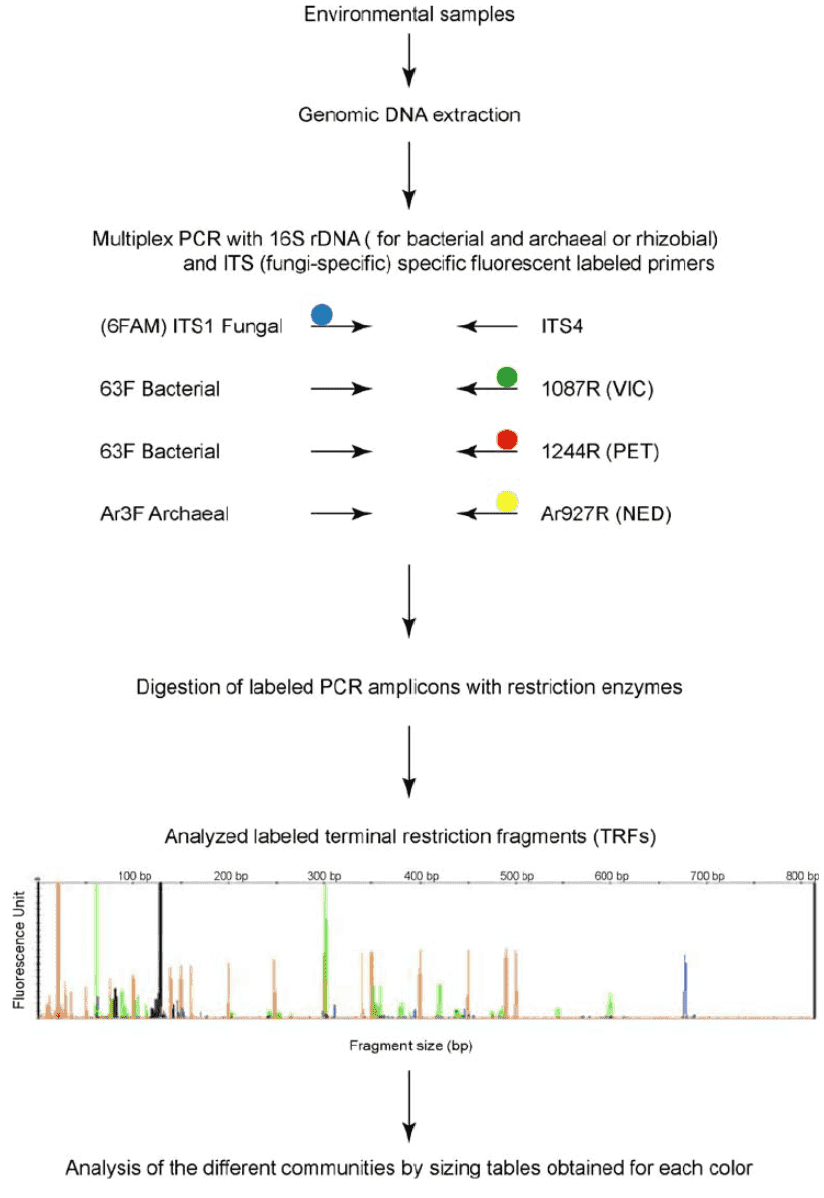
Sequencing of 16S/18S/ITS rRNA
Ribosomal RNA (rRNA) sequencing 16S, 18S and Internal Transcribed Spacer (ITS) are standard techniques of amplicon sequencing used to analyze and classify bacteria or fungi present within a particular set and can define strains that cannot be discovered using conventional methods.
Shotgun metagenomic sequencing can identify very low abundance members of the microbial community that can be skipped with other techniques with the ability to blend many microbial sequencing specimens in a single run and require maximum sequence coverage per specimen.
Microbial Transcriptome Analysis
Microbial RNA sequencing allows non-biased strand-specific detection of popular and novel transcripts, as opposed to hybridization-based techniques such as microarrays.
Whole-Genome Microbial Sequencing
Whole-genome sequencing (WGS) based on NGS helps scientists in microbiology to process hundreds of organisms with the power of multiplexing. No labor-intensive cloning steps are required, unlike traditional methods.
Case Study
Case 1
In diverse systems, the rapid growth of genomic techniques has dramatically improved the knowledge of microbial communities. GeoChip is a complete functional gene array that is composed of 24,243 oligonucleotide probes which cover 10,000 genes in 150 functional groups associated with nitrogen, carbon, and sulfur cycling, metal compression and protection, and organic contaminant degradation. It was established in order to truly comprehend the diversity and functions of microbial diversity. The GeoChip for the investigation of biogeochemical, ecological, and environmental processes has been shown to be a high-throughput and formidable genomic technology. In order to fulfill the contaminant composition, GC/MS and column chromatography were utilized and GeoChip was used to classify the modifications in the microbial functional gene after ozonation and in subsequent biodegradation treatments with and without bioaugmentation.
Case 2
It is essential for bioremediation of oil-contaminated soils to recognize the microbial functional diversity and the factors influencing microbial functions. However, it is hard to evaluate microbial functional structures quickly and comprehensively using conventional approaches to molecular ecology. Therefore, large-scale sequencing and/or microarray-based metagenomics tools for high-throughput are required. A functional gene array, GeoChip is a powerful metagenomics technology for the functional structure of microbial community analysis. Several studies have shown that GeoChip is an ideal method for analyzing the functional structure of the microbial community in both natural and contaminated conditions.
Case 3
In determining the greenhouse gas feedback of tundra ecosystems to the climate, the microbial degradation of soil organic carbon (SOC) in defrosting Arctic permafrost is essential. The structural changes of the microbial community during SOC decomposition, however, are poorly known. The alterations were studied in anoxic microcosm incubation at −2 and 8 ° C for 122 days using frozen soils from Barrow, Alaska, USA. To assess the microbial diversity and the functional genes associated with SOC degradation, methanogenesis, and Fe(III) reduction, the functional gene array GeoChip was used.
References
- Yang Z, Yang S, Van Nostrand JD, et al. Microbial community and functional gene changes in Arctic tundra soils in a microcosm warming experiment. Frontiers in microbiology. 2017(8).
- Liang Y, Van Nostrand JD, Deng Y, et al. Functional gene diversity of soil microbial communities from five oil-contaminated fields in China. The ISME journal. 2011, 5(3).
- Liang Y, Van Nostrand JD, Wang J, et al. Microarray-based functional gene analysis of soil microbial communities during ozonation and biodegradation of crude oil. Chemosphere. 2009, 75(2).