Cancer Transcriptomics Sequencing Promotes Drug Development
Among numerous diseases, cancer is a set of diseases characterized by unchecked cell proliferation and invasion of surrounding tissues and is the leading threat to modern human health. Surgery, radiotherapy, and chemotherapy are the three major clinical measures to treat cancer effectively. Chemotherapy, however, produces serious adverse effects, and chemotherapy drugs also badly damage immune function. In the face of the limited number of anti-cancer drugs on the global market and the increasing clinical needs of tumor drugs that have not been met, it is very necessary to targeted therapies for cancer and searches for new efficacy anticancer drug. Nevertheless, the sequencing technologies provide a vast catalogue of methodologies and databases required to analyze, integrate and interpret cancer multi-omics data. Remarkably, transcriptomics has driven the discovery and development of new anticancer drugs.
Along with advancements in high-throughput RNA sequencing (RNA-seq) and the development of computational algorithms, transcriptional diversity has been largely brought to light in human diseases. Transcriptional variants generate specific RNA transcripts that may play important roles in tumor development. For example, transcriptome analysis reveals the full information about differentially expressed genes in different treatments from specific cell types or tissues. There have been significant advances in precision oncology, with increasing adoption of sequencing tests that identify targetable mutations in cancer driver genes and recent studies have begun to explore the utilization of transcriptomics data to guide cancer patients' treatment. The data from this analysis can be used to characterize anti-cancer compounds and drug development.
Moreover, as an effective and efficient technology, it is hopeful to expound regulatory mechanisms of cancer cells are associated with various signaling pathways and seek differentially expressed genes to identify the molecular mechanisms and anti-cancer compounds involved to help develop natural cancer treatment drugs.
High-throughput transcriptome sequencing promotes to screen natural breast cancer treatment drugs
Breast cancer is one of the most commonly diagnosed malignant tumors among females. Research to develop new drugs for breast cancer treatment has focused on finding natural ingredients with low toxicity and high efficiency from TCM. Comparative transcriptome analysis is conducive to characterize anti-cancer compounds and drug development. A study used high-throughput transcriptome sequencing to provide the transcriptome data of breast cancer cell line distilled with 74 TCM and 10 chemical compounds. By comparing the changes in the gene expression among the cancer-related pathway of cell lines treated with TCM or chemical compounds with a known anti-cancer effect, three new TCM have been screened out that have the potential to develop natural drugs for breast cancer therapy, and obtained the therapeutic targets.
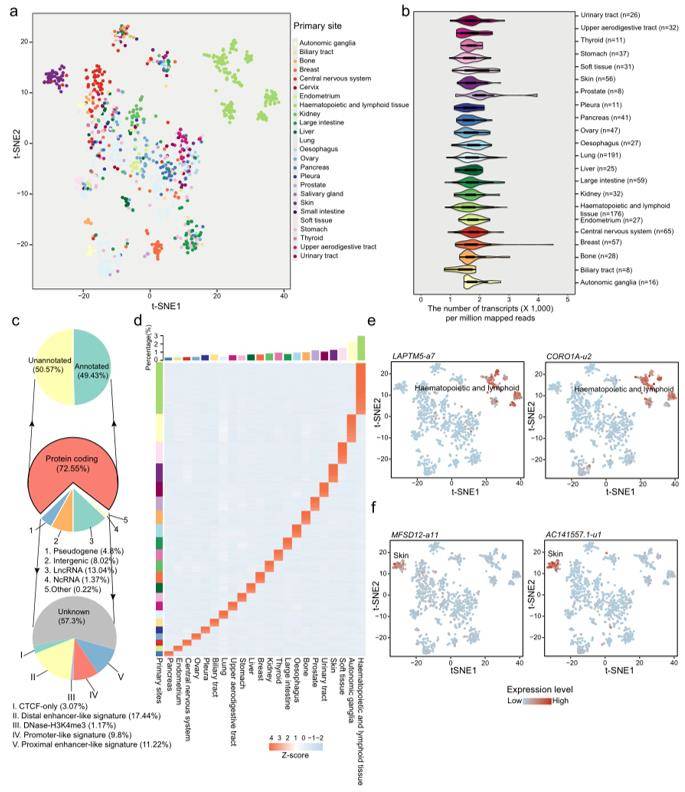 Transcriptional atlas across pan-cancer cell lines (Hu W et al., 2022)
Transcriptional atlas across pan-cancer cell lines (Hu W et al., 2022)
Transcript-level analysis provides potential treatment strategies in cancer
Human cancer-derived cell lines have been widely used as pre-clinical cancer models in cancer biology research and anti-cancer drug discovery. Researchers carried out a reference-based transcript assembly with RNA-seq data across more than 1000 cancer cell lines. Furthermore, RBP-transcript regulation and transcript-drug associations in cancer were combined to build RBP-transcript-drug axes, wherein PTBP1 is experimentally validated to affect the sensitivity to decitabine by regulating KIAA1522-a6 transcript. This study substantially extends cancer RNA repository, which facilitates anti-cancer drug discovery and the understanding of cancer transcriptome diversity and potential clinical utility at the transcript level.
Single-Nucleus RNA Sequencing contributes to developing clinical medication
Single-cell sequencing and spatial transcriptomics are state-of-the-art tools to unravel the cell heterogeneity and microenvironment of tumors. The aforesaid sequencing technology reveals the immunological microenvironment of cervical squamous cell carcinoma (CSCC) and identifies pro-tumorigenic cancer-associated myofibroblasts (myCAFs), which are expected to be a potential target for the treatment of CSCC. Collectively, snRNA-seq sheds light on the treatment and scientifically guides clinical drug development of advanced CSCC.
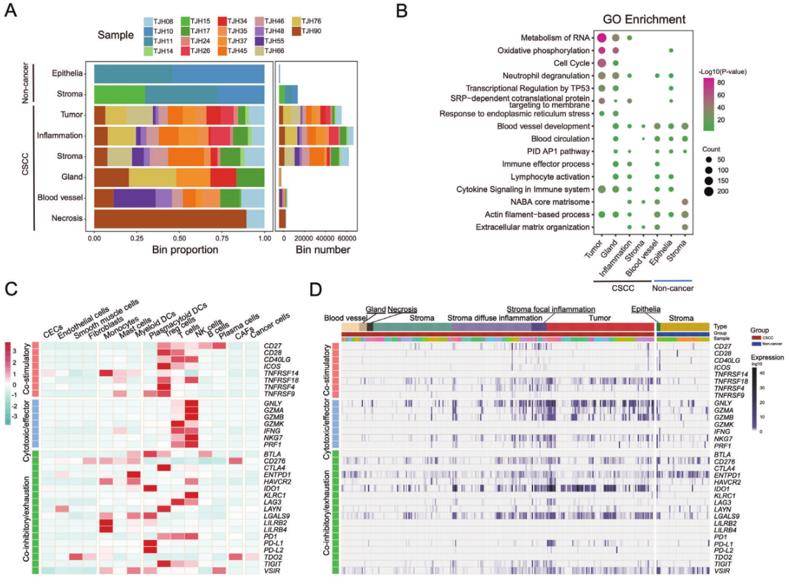 Transcriptomic analysis of the immunity and energy metabolism heterogeneity in cervical squamous cell carcinoma (CSCC) (Ou Z et al., 2022)
Transcriptomic analysis of the immunity and energy metabolism heterogeneity in cervical squamous cell carcinoma (CSCC) (Ou Z et al., 2022)
Summary
Utilization of transcriptomics to reveal molecular networks and screen drug targets has been proven to be a next-generation treatment for various cancers. Transcriptome has irreplaceable application potential in cancer from diverse aspects. In the future, the application of transcriptomics to direct personalized and precise drug use of this emerging technology will continue to be widely used and provide new insights into development and research of new cancer drugs.
References:
- Kui L, Kong Q, Yang X, et al. High-Throughput In Vitro Gene Expression Profile to Screen of Natural Herbals for Breast Cancer Treatment. Front Oncol. 2021; 11:684351. Published 2021 Aug 13.
- Hu W, Wu Y, Shi Q, et al. Systematic characterization of cancer transcriptome at transcript resolution. Nat Commun. 2022;13(1):6803. Published 2022 Nov 10.
- Ou Z, Lin S, Qiu J, et al. Single-Nucleus RNA Sequencing and Spatial Transcriptomics Reveal the Immunological Microenvironment of Cervical Squamous Cell Carcinoma. Adv Sci (Weinh). 2022;9(29): e2203040.


 Sample Submission Guidelines
Sample Submission Guidelines
