Integration Site Sequencing of Viral Gene Delivery Vehicles: Introduction, Sequencing Technology, and Applications
Inquiry >Overview of Viral Vectors
The introduction of specific cell function-altering genetic material into a patient to treat a genetic disease is known as gene therapy. The efficient delivery of genes to the target tissue/cells, which is carried out by gene delivery vehicles known as vectors, is a critical step in gene therapy. Viruses and non-viral vectors are the two types of vectors. In vivo delivery of the therapeutic gene into the patient using vectors based on retroviruses, adenoviruses (Ads), or adeno-associated viruses is the goal of today's viral vector-based gene therapy (AAVs).
Integrating and non-integrating viral vectors are the two types of viral vectors. The former, such as retroviral, lentiviral, and adeno-associated viral vectors, can incorporate into the human genome, whereas the non-integrating vector is kept in the nucleus but does not incorporate into chromosomal DNA, causing the transgene to be lost during cell division and the foreign gene's expression to be transient. The critical elements for furthering viral propagation can be provided in trans in a packaging cell, allowing viral vectors to be packaged as viral particles and deliver genes to the targeted cells. This is unquestionably a dead-end infection, as the vectors lack the necessary components for virus propagation. Infectious parental viruses can be produced using recombinant viral vectors. This is a principle that is frequently used in the design of viral vectors for gene therapy.
Applications of Retroviral Vectors
Preclinical and clinical applications of retroviral vectors are common. Retroviral vectors, unlike many other types of vectors currently being developed for gene therapy, can genetically modify cells in a stable manner without disrupting cell growth. Retroviral vectors based on murine retroviruses are ideal for ex vivo applications involving rapidly dividing cells. Retroviral viral vectors have been utilized in a variety of ex vivo gene therapy approaches for treating genetic diseases like Gaucher disease and severe combined immunodeficiency (SCID), as well as acquired diseases like cancer and arthritis.
Methods for Sequencing Analysis of Integration Sites
Retroviruses are one of the most important components of gene therapy approaches for treating diseases and correcting defective genes. The study of integration sites during gene therapy allows for the tracking of potential side effects and the development of safer integrating vectors. The risks associated with viral vector insertional mutagenesis have been reassessed considering adverse events reported from the X-linked severe combined immunodeficiency (X-SCID) gene therapy and other trials. As a result, elucidating vector insertion sites is critical for gene therapy applications in the future.
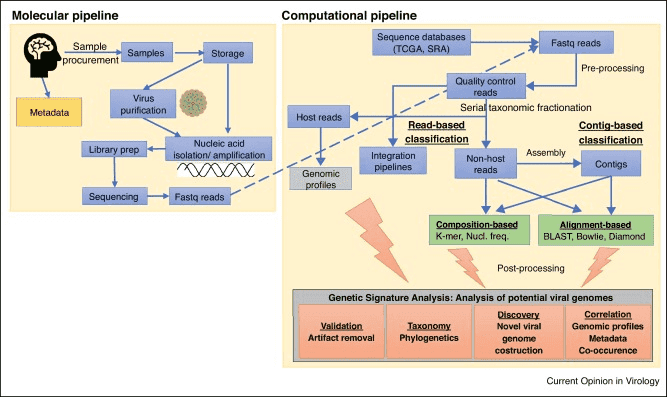
At vector integration sites, numerous efforts have been made to detect and sequence unknown DNA flanking sequences. The majority of these efforts rely on PCR amplification of target sites. Furthermore, using next-generation sequencing technologies, a large-scale analysis of vector integration sites has begun.
Targeted sequence capture of unique regions is a novel technique that can be applied to a variety of genomic applications. Sequence capture enrichment is an amplification-free method that hybridizes the region of interest with labeled, complementary DNA probes, and the DNA that has been captured is then sequenced. Lentiviral vector integration sites can be captured using targeted sequence capture technology. Thus, lentiviral vectors can be better applied with improved safety properties verified by NGS technology, which makes the process accurate, time-saving, and cost-effective.
References
- Bulcha JT, Wang Y, Ma H, et al. Viral vector platforms within the gene therapy landscape. Signal Transduction and Targeted Therapy. 2021;6(1).
- Mali S. Delivery systems for gene therapy. Indian journal of human genetics. 2013;19(1).
- Ustek D, Sirma S, Gumus E, et al. A genome-wide analysis of lentivector integration sites using targeted sequence capture and next generation sequencing technology. Infection, Genetics and Evolution. 2012;12(7).