Water shortage areas are moving to alternative drinking water supplies, including wastewater that has been purified to meet environmental requirements for potable water by enhanced treatment. Advanced treatment is accompanied by secondary or tertiary treatment of wastewater which usually requires MF and RO, followed by advanced oxidation (UV / H2O2) to degrade residual organic pollutants and microbial pathogens. Purified water is mixed into the drinking water delivery system indirect potable reuse systems after a period of storage, so microbial threats must be carefully addressed. Present laws center on the elimination of viruses and protozoan cysts and ensure that the risk level for these contaminants is as minimal as for conventional drinking water in advanced treated water. In order to explain how advanced treatment impacts the consistency of microbial water, the removal and growth of bacteria through treatment trains and delivery should be investigated.
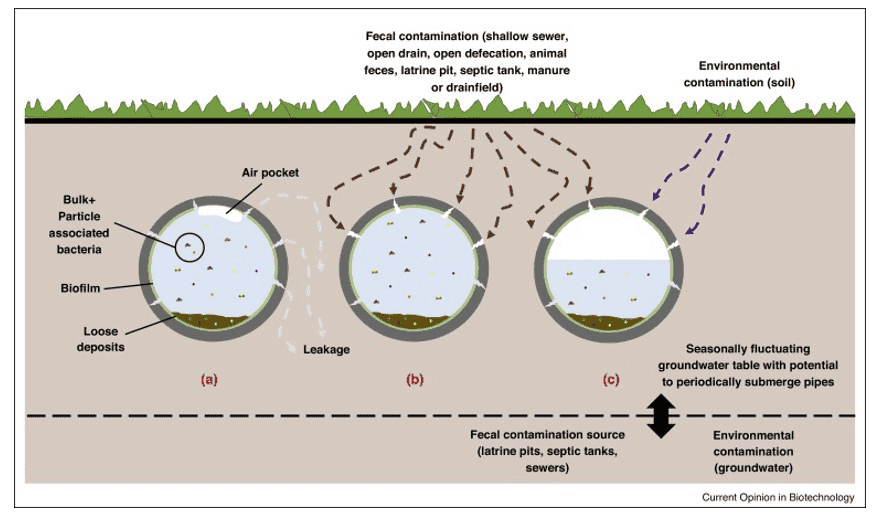 Figure 1. The impacts of intermittent supply on the drinking water microbiome. (Santos, 2019)
Figure 1. The impacts of intermittent supply on the drinking water microbiome. (Santos, 2019)
The aim of sampling is to capture a portion of material small enough in volume to be shipped easily and still sufficient enough for detection purposes while still reflecting the collected material correctly. This goal ensures that the relative quantities or concentrations of all related materials will be the same in the samples as in the collected content and that the sample will be treated in such a manner that no major differences in composition exist until the sample is sampled.
Sampling Method
Determine the concentration that satisfies the sampling software criteria and treat it such that when it is tested, it does not deteriorate or become tainted or corrupted. Load sample containers with a sample without pre-rinsing; pre-rinsing results in the removal of some pre-added preservative and often high bias results as such materials stick to the container sides. If a bottle already contains a preservative, be careful not to overfill the bottle, since it can remove or dilute the preservative. Since certain constituents can be present at low concentrations (micrograms or nanograms per liter), where adequate sampling and processing systems are not filled, they can be fully or partly lost or easily polluted.
Observed findings can differ with depth, streamflow, and distance from each shore when samples are obtained from a river or stream. The choice of the number and distribution of locations where samples should be obtained depends on the objectives of the analysis, the characteristics of the stream, the equipment available, and other considerations. Take an articulated sample from top to bottom in the middle of the mainstream channel or from side to side at mid-depth, if the material is available. If samples can be obtained only by grab or pick, preferably carry them across the stream at different points of equal distance; if only one sample can be collected, take it in the middle of the mainstream channel and at mid-depth.
There are major differences from natural sources, such as seasonal stratification, diurnal variations, rainfall, runoff, and wind, in rivers, streams, lakes, and reservoirs. Depending on the specific circumstances and the objective of the inquiry, choose the location, depth, and frequency of sampling. Limit regions with intense turbulence due to the likely loss of volatile materials and the potential existence of poisonous vapors that are denser than air. If necessary, prevent sampling at weirs because such sites appear to prefer extraction of lighter-than-water, insoluble substances. In general, in calm environments, gather samples under the surface and open a sampling jar below the surface with the mouth pointed at the current to prevent capturing surface scum unless oil and grease are an interesting component; then take water at the surface.
As sample components can be harmful, during collection and sample handling, take appropriate precautions. Toxic substances can penetrate through the skin and eyes, and also through the lungs in the case of vapors. Ingestion can happen by direct contact with food with hazardous chemicals or by vapor adsorption into food. Precautions may be confined to wearing hats, or coveralls, aprons or other protective garments may be included. Sample only in well-ventilated environments, or use an effective respirator or self-contained ventilation device where harmful vapors may be involved. Open sample containers in a fume hood in a laboratory. Keep fires and intense sources of heat away from samples and places of sampling. Using only specially built explosion-proof refrigerators where flammable substances are expected or proven to be present and specimens are to be kept in the fridge. Safely collect samples, preventing circumstances that can lead to incidents.
Sample Pretreatment
Remove organics to reduce possible spoilage of the resins in applications where the feedwater has large amounts of organic matter. Possible pretreatments involve adsorption or reverse osmosis.
Reverse osmosis is a mechanism in which a part of dissolved constituents and suspended impurities are separated by pressing water under pressure through a semipermeable membrane. Product water quality depends on the quality of the feedwater.
Adsorption is commonly used to dissolve chlorine and organic contaminants. Usually, for granular activated charcoal, it is done. The success of the removal of organic compounds depends on the quality of the organic pollutants, the functional properties of the activated carbon, and the conditions of activity. In general, the performance of organic adsorption is inversely proportional to solubility and may be insufficient to remove polar compounds of low molecular weight.
Reference
- Bautista-de los Santos QM, Chavarria KA, Nelson KL. Understanding the impacts of intermittent supply on the drinking water microbiome. Current opinion in biotechnology. 2019, 57:167-74.

