The oral microbiome is the second most diverse microbial community in the human body, next to the gut microbiome, harboring over 700 species of bacteria. The human oral cavity composed of different habitats, the teeth, tongue, cheeks, soft and hard palate, gingival sulcus, and tonsils. Each of these special habitats has a unique composition of microorganisms as some microbes are more suited to soft oral tissues and others to hard tissues such as teeth. Some of the important factors affecting the microbial population in the oral cavity include high moisture, rich in nutrients, and moderate temperature.
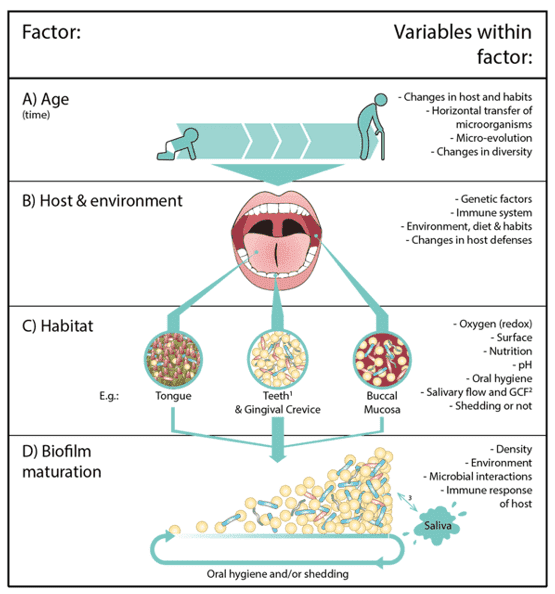 Figure 1. Factors that determine the composition of the oral microbiota. (Rosier, 2018)
Figure 1. Factors that determine the composition of the oral microbiota. (Rosier, 2018)
Oral microbes play an important role in functions such as metabolism, maintenance of teeth structure, and even in the regulation of blood pressure. Also, are crucial in maintaining oral and systemic health since the mouth is the initiation point of digestion. These microbes specifically protect the mouth against colonization of extrinsic bacteria which might affect systemic health.
The oral microbiome has been extensively explored with cultivation and culture-independent molecular methods such as NGS-based amplicon sequencing. To date, the oral cavity is one of the most explored human microbiomes as the emergence of new genomic technologies, particularly next-generation sequencing, and bioinformatics, revealed its complexities.
Considerations Before Sampling
Subjects are screened prior to the sampling and there are several conditions that may result in their exclusion. These include chronic dry mouth, with periodontal pockets less than four millimeters, and untreated carious lesions or oral abscesses. The subject may also be excluded if she/he has evidence of precancerous or cancerous lesions, candidiasis, and clinically meaningful halitosis. Lastly, if the subject has more than eight missing teeth and has more than 10% of sites of bleeding on probing.
Sampling Method
The aseptic technique must be used throughout the collection of all specimens. The sequence of sampling is the collection of saliva, followed by collection on soft tissue sites, and then collection on hard tissue sites. Since sampling on the throat is difficult due to gag reflex, it is suggested that sampling in this area must be done after sampling on the other mentioned sites.
Saliva
The subject will be asked to collect her own saliva for at least one minute and drool into the labeled MicroCollect™ Saliva Collection Device. This specific process might be done several times to be able to collect a larger volume of saliva, about two to five milliliters. The tube containing saliva will be put in a Ziploc bag and place over ice. This must be accomplished within four hours prior to transfer to the clinical laboratory for processing.
Soft Tissue Sites
Sampling in these sites will be accomplished using MicroCollect™ Sterile Pack Swabs. To ensure the transfer of bacteria from the swan into the solution, the nylon fiber part of the swab must be pressed against the wall of the tube several times for about 20 seconds. The specimens must be kept cold prior to the processing.
Hard Tissue Sites
Supragingival Plaque: The site to be sampled must first be isolated using cotton rolls and dry with a gentle stream of air using an air-water syringe. Remove all the supragingival plaque from the mesial surface of the selected index tooth using a Gracey curette. The tip of the curette must be immersed in buffer for about 4-5 seconds. Dry the face of the curette on the inside edge of the MicroCollect™ Oral Collection and Preservation Device. Repeat the process on each index tooth. The specimens must be kept cold prior to the processing.
Subgingival Plaque: Place the cotton rolls, dry the area, and remove any supragingival plaque residuals prior to the collection of subgingival plaque. Using a sterile Gracey curette, collect the subgingival plaque from the mesiobuccal surface of the selected index tooth. The tip of the curette must be immersed in buffer for about 4-5 seconds. Dry the face of the curette on the inside edge of the collection device. Re-sampling of the same site is not recommended due to the risk of bleeding. The specimens must be kept cold prior to the processing.
References
- Deo PN, Deshmukh R. Oral microbiome: Unveiling the fundamentals. Journal of oral and maxillofacial pathology. 2019, 23(1):122.
- Rosier BT, Marsh PD, Mira A. Resilience of the oral microbiota in health: mechanisms that prevent dysbiosis. Journal of dental research. 2018, 97(4):371-80.
- Arweiler NB, Netuschil L. The oral microbiota. Microbiota of the Human Body. Springer, Cham. 2016, 45-60.
- Dewhirst FE, Chen T, Izard J, et al. The human oral microbiome. Journal of bacteriology. 2010, 1;192(19):5002-17.

