The gut microbiota portrays a specific microbial community and has been identified with great significance towards human health. Environmental variables and dietary habits could affect the composition and function of gut microbiota. Control of the intestinal microbiota by dietary treatments, pre-or probiotic treatment, and the success of faecal microbiota transplants (FMT) may now be regarded as a therapeutic application.
Faeces are harvested in a clear, non-invasive manner and appear to have a very high density of microbes. The amount of bacteria in the faeces, however, relies on the purity of the faeces. Therefore, it is best to replicate faecal sampling over time to further grasp the complexity of the intestinal microbiota of an organism, as well as its modifications and tolerance. It is hard to maintain the identity of gut microbiota in faecal samples and small adjustments during sample handling can yield great variation in subsequent outcomes. Therefore, during the pretreatment of faeces, pre-analytical preparation should include protection procedures.
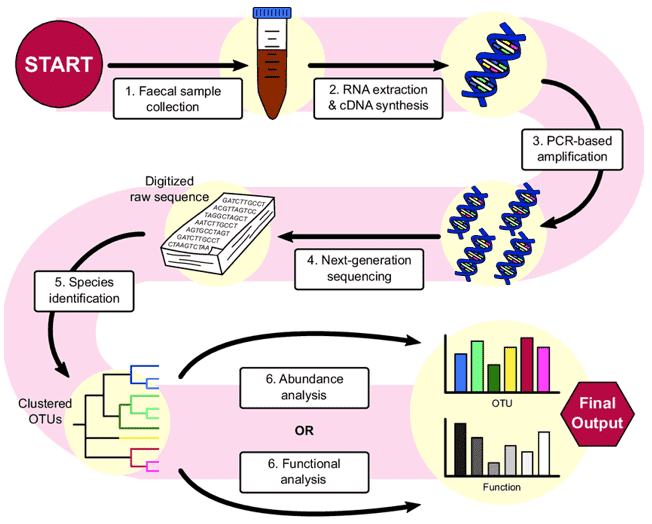 Figure 1. From stool to statistics. Overview of a sample method used to analyze the gut microbiome using 16S sequencing, a popular technique in microbiome research. (Bastiaanssen, 2019)
Figure 1. From stool to statistics. Overview of a sample method used to analyze the gut microbiome using 16S sequencing, a popular technique in microbiome research. (Bastiaanssen, 2019)
Sampling Method
Preferably, collecting faeces should be done immediately to eliminate variations of temperature, and aseptic states should be retained during processing. Two main features must be fulfilled by the collection kit: guaranteeing that appropriate temperature conditions are maintained and avoiding contamination. Meanwhile, reduced stool firmness has been described to be significantly related to reduced faecal bacteria richness and disturbed microbial communities. Generally, the kit includes a hermetic bag, a container and lid for stool collection, collection paper for stool collection, a pair of gloves, and sample coding and tracking labels. Depending on the method employed, the quantity may differ. Indeed, with all omic methods, the volume of sample extracted and the manner in which it is stored would not be the same. For example, more sample amounts will be needed for metabolomic analysis, whereas a metagenomic or transcriptomic analysis will require the inclusion of some reagents. It should be frozen as soon as practicable after the sample has been processed, as there may be a substantial decrease in Bacteroidetes phylum and Firmicutes phylum rise at room temperature after 30 minutes of storage.
Transport and Storage
Faeces should be frozen for less than three days in the freezer (approximately −20 °C) for optimum transport to the lab where they should be kept at −80 °C. The specimen should be taken as soon as possible to the final storage location and temperature variations should be prevented, as unexpected changes in temperature can induce changes in the bacterial function and composition of the sample, creating distress in the bacteria in the faeces. To stop the thawing phase and sample degradation, transport needs to be as quick as possible and the temperature should be regulated and not higher than 4 °C. Cold bag or preferably dry ice is required for cold transport to be carried out. Since up to 4 freeze-thaw cycles can be carried out with no difference in the microbial composition, take temperature as the key reference. However, changes in the faecal metabolome that may be related to the release of intracellular material after cell lysis due to the development of crystals that affect the cell wall during freezing. To prevent freeze-thaw periods and to ensure the least practicable modification of the faecal sample, preparing sample recording, aliquoting and pre-treatment is therefore important.
Sample Pre-Treatment
The sample homogenization prior to all other pretreatment and aliquoting would give the entire faeces a representative microbial population composition and a representative metabolite profile. In addition, since there is high intra-individual variability in faecal samples, it is suggested that a reasonably significant volume of stool should be obtained. Before freezing, the second possible pretreatment is to retrieve the faecal water because freezing could boost BCAAs, aromatic amino acids, intermediates of the Krebs cycle and monosaccharides, and decrease SCFAs, such as butyrate. Lyophilization is the third potential pretreatment. Due to inter-individual variations in quality, lyophilization is conducted on crude faecal samples. On the positive side, it allows specimens to be standardized, using their dry weight and retaining microbial and chemical stability. Finally, the separation of metabolites from the sample must be carried out, regardless of the pre-treatment conducted.
References
- Zubeldia-Varela E, Barber D, Barbas C, et al. Sample pre-treatment procedures for the omics analysis of human gut microbiota: turning points, tips and tricks for gene sequencing and metabolomics. Journal of Pharmaceutical and Biomedical Analysis. 2020, 28:113592.
- Bastiaanssen TF, Cowan CS, Claesson MJ, et al. Making sense of… the microbiome in psychiatry. International Journal of Neuropsychopharmacology. 2019, 22(1):37-52.
- James SC, Fraser K, Young W, et al. Gut microbial metabolites and biochemical pathways involved in irritable bowel syndrome: Effects of diet and nutrition on the microbiome. The Journal of Nutrition. 2020, 1;150(5):1012-21.
- Gratton J, Phetcharaburanin J, Mullish BH, et al. Optimized sample handling strategy for metabolic profiling of human feces. Analytical chemistry. 2016, 3;88(9):4661-8.

