Introduction
Malignant tumors derived from mesenchymal tissue (including connective tissue and muscle) are called "sarcomas". Sarcoma accounts for 12.6% of all malignant tumors in children and adolescents under 19 years of age but is rare in adults. Sarcoma is divided into two categories, soft tissue sarcomas which occupies the majority of sarcoma and malignant primary bone tumors which occupies approximately 10%. The clinical manifestation of sarcoma includes local pain and swelling, or local skin redness, heat, joint effusion, limb or joint activity pain and restriction, etc. The etiology of sarcoma is unknown and the pathogenesis is unclear. However, it has been reported that mutations in some genes are associated with increased risk of sarcoma.
Disease-related gene description
The mutations in TP53, RB1, NF1, SDHA, SDHB, SDHC and SDHD genes are frequently detected in sarcomas. The tumor suppressor gene TP53 is the most common mutated gene in human cancer, which also increases the risk of other cancers such as melanoma, breast cancer and thyroid cancer. In sporadic cancers, the prevalence of TP53 variants in somatic cells is from 10% to 60%. p53 is inactivated due to the missense mutation, thereby destroying a complete set of anti-proliferative and growth-inhibiting responses. The p53 protein mutant has carcinogenic effects and promotes tumor development. RB1, also the tumor suppressor gene, plays a major role in the formation of sarcoma. Significant enrichment of pathogenic variation is reported in ATM and ATR genes which are related to DNA damage-sensing. Germline mutations in mitochondrial complex II succinate dehydrogenase (SDH) enzyme are commonly detected in gastrointestinal stromal tumors. In addition to these genes, the unexpected pathogenic variants in ERCC2 are confirmed, which can affect DNA binding, DNA damage sensing, helices activity and basal transcription.
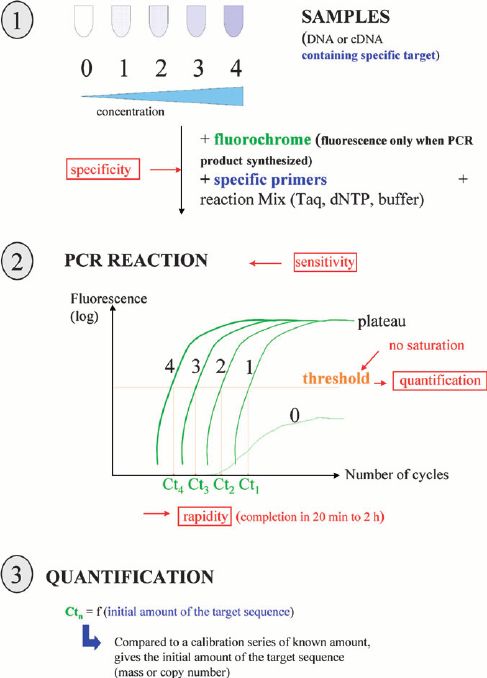
CD-Genomics provides a custom sarcoma panel containing optimized genes that are reported associated with the increase of risk of sarcoma. You can select the genes only you require to customize your exclusive panel. Targeted DNA sequencing by Illumina MiSeq system/Ion PGM system is provided to investigate the genetic variations for sarcoma seen in routine clinical practice.
Custom sarcoma panel offers but are not limited to:
-
Targeted DNA sequencing technology by Illumina MiSeq system/Ion PGM system provides ultra-deep sequencing for target specific genomic regions, so that we can detect low frequency variants of sarcoma.
-
Identifying multiple sarcoma-related genes in a single assay is rapid and convenient.
-
Strict quality control throughout the pipeline workflow ensures the accuracy and repeatability of the sequencing.
-
Every detected genetic variant will be further validated to ensure the validity of results.
-
Custom panel content is designed to keep up with the frontiers from current literature about the sarcoma panel to target all relevant regions.
-
Precision bioinformatics pipelines ensure superior analytical performance.
Choose the genes that suit you from the sarcoma gene list
| ALK |
APC |
ATM |
ATR |
BCOR |
| BLM |
CAMTA1 |
CCNB3 |
CDKN1C |
CIC |
| DICER1 |
EPC1 |
EPCAM |
ERCC2 |
EWSR1 |
| EXT1 |
EXT2 |
FH |
FOS |
FOSB |
| FOXO1 |
FUS |
GLI1 |
HMGA2 |
HRAS |
| JAZF1 |
MEAF6 |
MKL2 |
NCOA2 |
NTRK1 |
| NTRK2 |
NTRK3 |
NUTM1 |
PAX3 |
PDGFB |
| PDGFRA |
PLAG1 |
PMS2 |
PTCH1 |
RB1 |
| ROS1 |
SS18 |
STAT6 |
TAF15 |
TCF12 |
| TFE3 |
TFG |
TP53 |
USP6 |
YWHAE |
Specimen requirements of our custom sarcoma panel
- Specimen: blood, saliva or extracted DNA.
- Volume: 2-5 mL blood, 2 mL saliva and 3ug DNA.
- Collection: blood is collected by routine blood collection and saliva is collected by saliva collection kits (kits are available upon request). DNA samples are stored in TE buffer or equivalent.
- Container: lavender-top (EDTA) tube or yellow-top (ACD) tube.
- Storage/transport temperature: room temperature.
Gene panel workflow
For more information about the Custom Sarcoma Panel or need other amplification requirements, please contact us.
References:
- Ognjanovic S, et al. Sarcomas in TP53 germline mutation carriers: a review of the IARC TP53 database. Cancer, 2012, 118(5): 1387-1396.
- Ballinger M L, et al. Monogenic and polygenic determinants of sarcoma risk: an international genetic study. The Lancet Oncology, 2016, 17(9): 1261-1271.
- Stratakis C A, Carney J A. The triad of paragangliomas, gastric stromal tumours and pulmonary chondromas (Carney triad), and the dyad of paragangliomas and gastric stromal sarcomas (Carney–Stratakis syndrome): molecular genetics and clinical implications. Journal of internal medicine, 2009, 266(1): 43-52.
- Praja D I, et al. Association Between Patient Characteristics and Diet Profile with Kirsten rat sarcoma (KRAS) and Neuroblastoma rat sarcoma (NRAS) Gene Mutation in Colorectal Cancer. The Indonesian Journal of Gastroenterology, Hepatology, and Digestive Endoscopy, 2019, 20(1): 2-6.
* For research purposes only, not intended for clinical diagnosis, treatment, or individual health assessments.
Related Services
Related Products
Related Resources