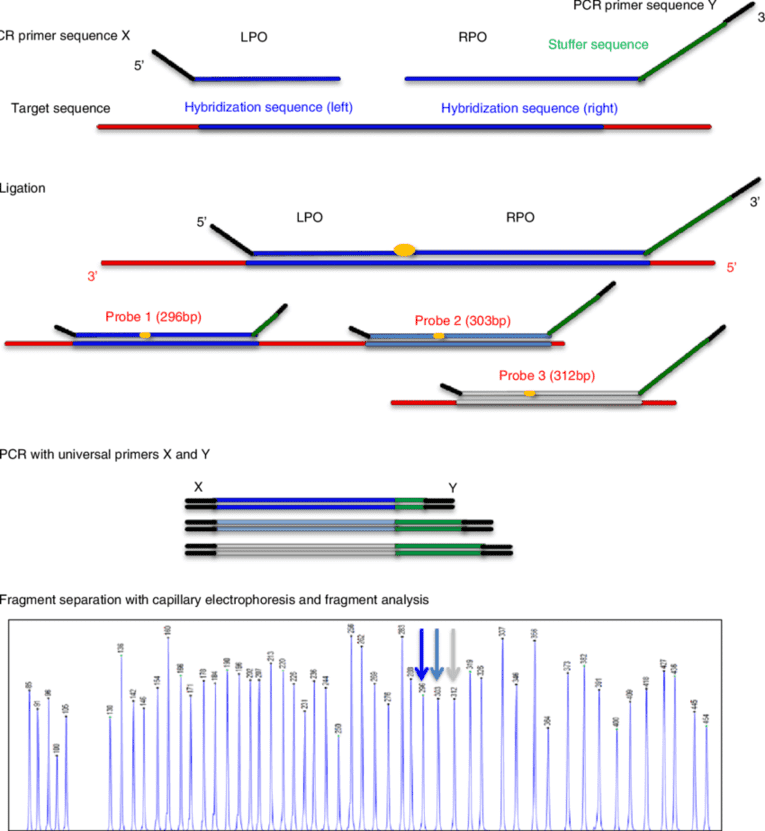
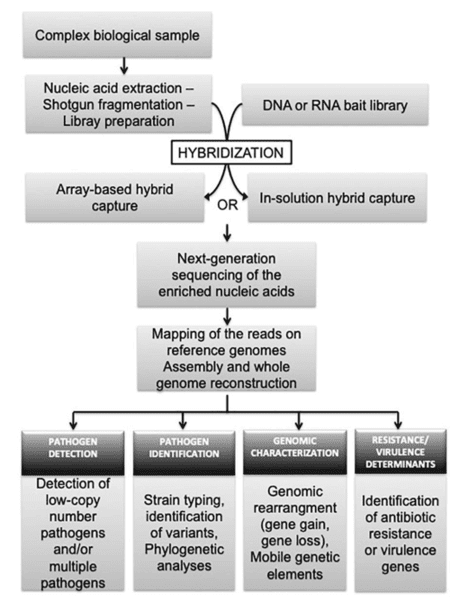
Custom MDS/MPN Overlap Panel
Introduction
Myelodysplastic/myeloproliferative neoplasms (MDS/MPN) are clonal myeloid disorders combining dysplastic and proliferative features of both myelodysplastic syndromes MDS and myeloproliferative neoplasms (MPN), but does not belong to these two categories. MDS/MPN carries more gene mutations associated with activation of growth factor signaling pathways, epigenetic regulatory and morphological abnormalities compared to MDS and MPN. MDS/MPN is currently divided into five types, chronic myelomonocytic leukemia (CMML), atypical chronic myeloid leukemia BCR-ABL1 negative (aCML), juvenile myelomonocytic leukemia (JMML), MDS/MPN with ring sideroblasts and thrombocytosis (MDS/MPN-RS-T) and myelodysplastic/myeloproliferative neoplasm, unclassifiable (MDS/MPN-U).
Disease-related gene description
Mutation screening for MDS/MPN individuals reveals frequent mutations of RUNX1 and CEBPA. RUNX1 (also known as AML1 or CBFA2) is located on chromosome band 21q22.12 and encodes an alpha subunit of the core binding factor (CBF) complex. This complex activates and inhibits transcription of key regulatory factors in the growth, survival and differentiation pathways. RUNX1 has also been identified as one of the most common targets in leukemia and somatic mutant chromosomal translocations. CCAAT enhancer binding protein A (CEBPA) gene and its coding transcription factor CCAAT enhancer binding protein alpha (C/EBPα) play an important role in early differentiation of hematopoietic system by promoting granulation differentiation of hematopoietic stem/progenitor cells and inhibiting cell proliferation. Mutations in the CEBPA gene and the abnormal regulation of transcription, translation and posttranslational levels can cause the abnormal structure or expression of C/EBP α protein, leading to the dysplasia of granule-line differentiation and abnormal proliferation of immature granule-line precursor cells. Mutations in transcription and other nuclear factors are common in myelodysplastic/myeloproliferative tumors and are often mutually exclusive. The mutations of SF3B1, a gene encoding the core component of RNA splicing mechanism, are closely related to the disease phenotype of annular astrocytes in MDS/MPN. TET2 is widely expressed in hematopoietic cells, but its function is unknown. JAK2-V617F mutations have been detected in up to 60% of MDS/MPN-U populations.
To support researches related to MDS/MPN overlap associated genes, our custom MDS/MPN overlap panel platform offers a comprehensive MDS/MPN overlap panel library from which you can choose for genetic testing of MDS/MPN overlap disease. High-throughput amplicon sequencing technology that we perform can screen genetic variants among the MDS/MPN overlap gene panel via a high effective mode and help researchers decode the MDS/MPN biology and identify MDS/MPN subtype.
Custom MDS/MPN Overlap Panel offers but are not limited to:
-
Illumina MiSeq system/Ion PGM system-based high throughput amplicon sequencing provides the advantages as follows: unparalleled coverage uniformity, highly targeted resequencing and detection of low frequency MDS/MPN overlap variants.
-
Provide the customizable MDS/MPN overlap panel sequencing which meets your requirements and saves costs.
-
Each genetic variant detected will be further validated to ensure the validity and reliability of results.
-
Our platform pipeline workflow designs the strict quality control in order to ensure the accuracy and repeatability of the sequencing.
-
Custom MDS/MPN overlap panel content is designed to keep pace with the current frontiers from MDS/MPN overlap literature to target all relevant regions.
-
You can choose our MDS/MPN overlap panel library content or discuss your requirements about custom MDS/MPN overlap panel, then we can design your own panel according to your requirements.
Choose the genes that suit you from MDS/MPN overlap gene lists
| ASXL1 |
CALR |
CBL |
| CEBPA |
CSF3R |
DNMT3A |
| ETNK1 |
EZH2 |
FGFR1 |
| IDH1 |
IDH2 |
JAK2 |
| KIT |
KRAS |
MPL |
| NF1 |
NRAS |
PDGFRA |
| PDGFRB |
PTPN11 |
RUNX1 |
| SETBP1 |
SF3B1 |
SH2B3 |
| SRSF2 |
TET2 |
TP53 |
| U2AF1 |
|
|
Specimen requirements of our custom MDS/MPN overlap panel
- Specimen type: peripheral whole blood, bone marrow or extracted DNA (not FFPE-compatible).
- Specimen volume: 10 mL whole blood, 2 ml bone marrow, 1 μg DNA.
- Collection: blood is collected by routine blood collection and bone marrow is collected into the appropriate container. DNA samples are stored in TE buffer or equivalent.
- Collection container: EDTA tube (lavender-top) or ACD tube(yellow-top).
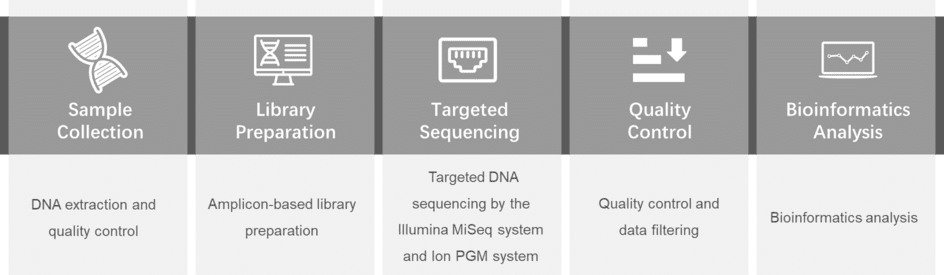
Gene panel workflow
Please contact us if you need more details about the Custom MDS/MPN Overlap Panel or other requirements.
References:
- Hyjek E1, Vardiman JW. Myelodysplastic/myeloproliferative neoplasms. Semin Diagn Pathol. 2011 ,28(4):283-97.
- Sakr R, et al. Next-generation sequencing discriminates myelodysplastic/myeloproliferative neoplasms from paraneoplastic leukemoid reaction in cancer patients with hyperleukocytosis. Leukemia & lymphoma, 2018, 59(7): 1742-1745.
- Malcovati L, et al. Clinical significance of SF3B1 mutations in myelodysplastic syndromes and myelodysplastic/myeloproliferative neoplasms. Blood, 2011, 118(24): 6239-6246.
- Tiu R V, Sekeres M A. Making sense of the myelodysplastic/myeloproliferative neoplasms overlap syndromes. Current opinion in hematology, 2014, 21(2): 131-140.
- Jankowska A M, et al. Loss of heterozygosity 4q24 and TET2 mutations associated with myelodysplastic/myeloproliferative neoplasms. Blood, 2009, 113(25): 6403-6410.
- DiNardo C D, et al. Myelodysplastic/myeloproliferative neoplasms, unclassifiable (MDS/MPN, U): natural history and clinical outcome by treatment strategy. Leukemia, 2014, 28(4): 958.
* For research purposes only, not intended for clinical diagnosis, treatment, or individual health assessments.
Related Services
Related Products
Related Resources