Introduction
Myeloproliferative neoplasms (MPN) are a group of diseases caused by late-onset chronic hematopoietic dysfunction, including up to 97% of polycythemia vera (PV), about half of essential thrombocythemia (ET) and primary myelofibrosis (PMF). Symptoms and signs commonly shared by these disorders represent the effects of the excess blood cells or their functional dysregulation. According to the 2016 World Health Organization (WHO) classification, PMF can be divided into prefibrotic primary myelofibrosis (pre-PMF) and overtly fibrotic primary myelofibrosis (ver-PMF).
Disease-related gene description
The discovery of JAK2V617F mutation, a nonreceptor tyrosine kinase, has shed new light on the diagnosis and identification of clinical diseases, and also shows a bright future for the development of targeted drugs. The frequency of JAK2V617F mutation varies among different cMPD. In the original patient series, the majority of patients with PV (65–97%) have the JAK2 V617F. In contrast, only 23–57% of ET patients and 35–57% of those with idiopathic myelofibrosis (IMF) are found positive for the JAK2 mutation. Except for JAK2V617F mutation, several molecular biomarkers of MPN have been found, including MPL, LNK, CBL, TET2, ASXL1, IDH, IKZF1, EZH2, DNMT3A, TP53, SF3B1, SRSF2, etc. Interestingly, it's has been showed that the presence of mutations in two or more of these five genes predicts the worst survival. Furthermore, mutations in TP53 and TET2 also correlate with worse prognosis and an increased risk of leukaemic transformation.
To develop more effective drugs in treating MPN through mutant genes related to MPN, we offer a comprehensive MPN panel library from which you can choose for genetic testing of MPN. We can use amplicon sequencing technology to efficiently validate genetic variants between the MPN mutant genes via Illumina MiSeq system/Ion PGM system. And based on the technical process, researchers could learn more about the principles and develop related drugs to treat MPN.
Custom core MPN panel offers but are not limited to:
-
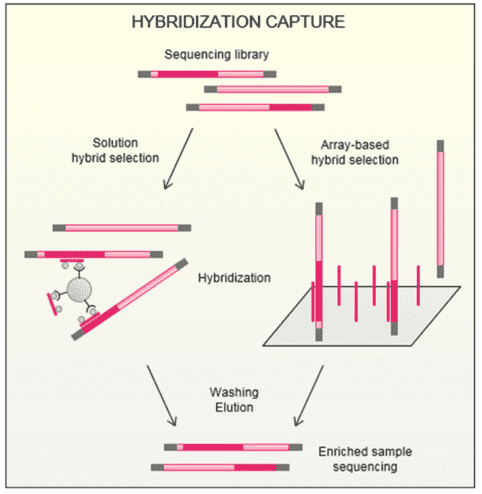
Amplicon sequencing technology by the Illumina MiSeq system/Ion PGM system is highly efficient and can be used to explore, validate and screen for MPN genetic mutation using a highly targeted approach.
-
Up to thousands of amplicons can be reused per reaction for high coverage.
-
Providing high-target resequencing is equally suitable for areas (such as areas with high GC content) that are difficult to sequence.
-
This technology is flexible and suitable for many types of experimental designs.
-
Every detected genetic variant will be further validated to ensure the validity of the results.
-
Using this method could reduce sequencing costs and turnaround time.
-
You can choose the panel content from our core MPN library to discuss your custom core MPN requirements, then we can provide you with your own panel.
Choose the genes that suit you from the core MPN panel gene list
| JAK2V617F |
MPL |
LNK |
CBL |
| TET2 |
ASXL1 |
IDH1 |
IKZF1 |
| EZH2 |
DNMT3A |
TP53 |
SF3B1 |
| SRSF2 |
CALR |
MPL515 |
TET2 |
| ASXL1 |
EZH2 |
MPL |
ASXL1 |
| U2AF1 |
GATA2 |
IDH2 |
PHF6 |
| NRAS |
SETBP1 |
BIOR |
BRAF |
| ETV6 |
KIT |
KRAS |
NPM1 |
| RUNX1 |
WT1 |
|
|
Specimen requirements of our custom core MPN panel
- Specimen: mononuclear bone marrow, peripheral blood cells or extracted DNA.
- Volume: 300 μL mononuclear bone marrow, 300 μL peripheral blood cells, 2 μL extracted DNA.
- Collection: mononuclear bone marrow blood and peripheral blood cells are collected by routine blood collection. DNA samples are stored in TE buffer or equivalent.
- Container: lavender-top (EDTA) tube or yellow-top (ACD) tube.
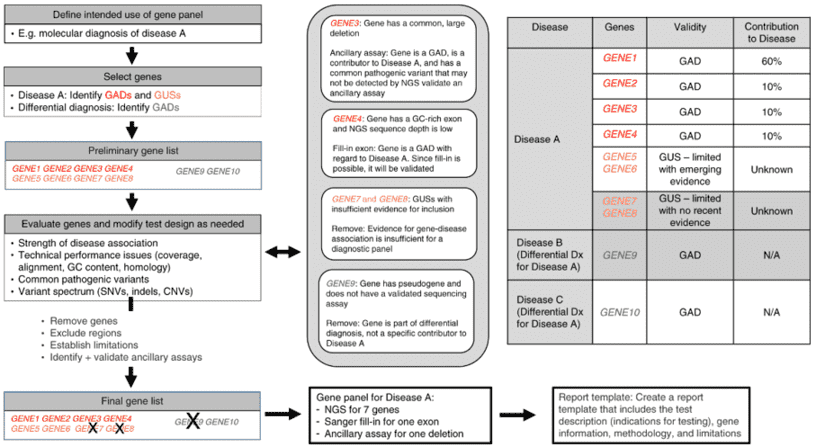
Gene panel workflow
For more information about the Custom Core MPN Panel or need other amplification requirements, please contact us.
Reference:
- Delic S, et al. Application of an NGS-based 28-gene panel in myeloproliferative neoplasms reveals distinct mutation patterns in essential thrombocythaemia, primary myelofibrosis and polycythaemia vera. British journal of haematology, 2016, 175(3): 419-426.
* For research purposes only, not intended for clinical diagnosis, treatment, or individual health assessments.
Related Services
Related Products
Related Resources