Bioinformatics Analysis for Preclinical Research
Preclinical research refers to the chemical synthesis or natural product purification research and drug analysis research conducted before the drug enters the clinical research, including pharmacodynamics, pharmacokinetics and toxicology research, as well as pharmaceutics research and drug evaluation of potential therapeutic interventions in cells and animals. Then, candidates for clinical trials are selected based on the effectiveness and safety of the drug in the disease model. Before the start of clinical trials, all drugs need data from various toxicological preclinical trials to study the safety of the drug in humans.
Application of Bioinformatics in the Preclinical Research
With the development of genomes, genomics methods are often used in the target identification/validation stage of drug development, but genomics methods are often ignored in the preclinical stage. Before the drug enters the clinic, it is necessary to conduct efficacy and safety tests in vivo and in vitro. This process is time-consuming and expensive. Moreover, the results of efficacy and safety evaluation data obtained are not representative of all individuals. Through genomics methods, microarray technology or next-generation sequencing technology, combined with bioinformatics methods, genomic changes related to toxicology or non-targeting can be effectively identified. It provides a data basis for subsequent wet experiments and saves time, manpower and material costs for pre-clinical research to a large extent.
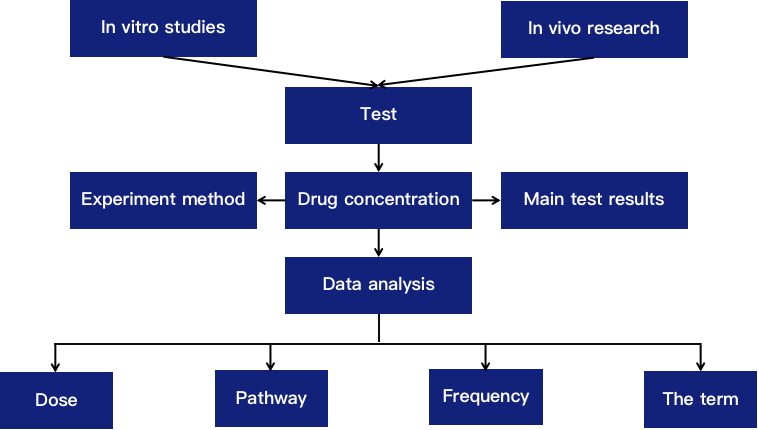 Fig 1. DDs (Doses of drugs) clustering of GMP and PPAR-SP datasets based on the Euclidean distance method in combination with the ward HC method. (Hasan MN, et al. 2019)
Fig 1. DDs (Doses of drugs) clustering of GMP and PPAR-SP datasets based on the Euclidean distance method in combination with the ward HC method. (Hasan MN, et al. 2019)
How We Can Help Your Preclinical Research
By combining our expertise in bioinformatics analysis with our extensive knowledge in the field of preclinical research, our bioinformatics service can help:
1. Genotyping analysis: by performing genotyping analysis on microarray data or sequencing data of various omics, mining genetic polymorphisms related to individual differences in drug response, including:
- Through the analysis of gene polymorphisms related to drug-metabolizing enzymes, predict the causes of individual differences in drug reactions.
- Analyze the genetic polymorphisms of drug transporters to predict the causes that affect drug transport, absorption, distribution, and elimination from the body.
- The analysis of gene polymorphisms of drug targets can predict the causes of the effects of different drugs (for example, different doses) in individuals.
2. Data mining: Combined with relevant drug databases, it can predict the efficacy of drugs, predict the dose of drugs, avoid ADR of drugs and find new targets for drugs.
In addition, if you have other analysis needs related to preclinical research, we will provide you with analysis services. We will use cutting-edge analysis technology to conduct personalized data analysis according to the customer's project analysis needs.
An Example of Preclinical Research Process
CD Genomics provides a one-stop bioinformatics data analysis service, and customers only need to provide raw data. We provide data analysis for your entire preclinical research data. The general pharmacodynamic test process is as follows:
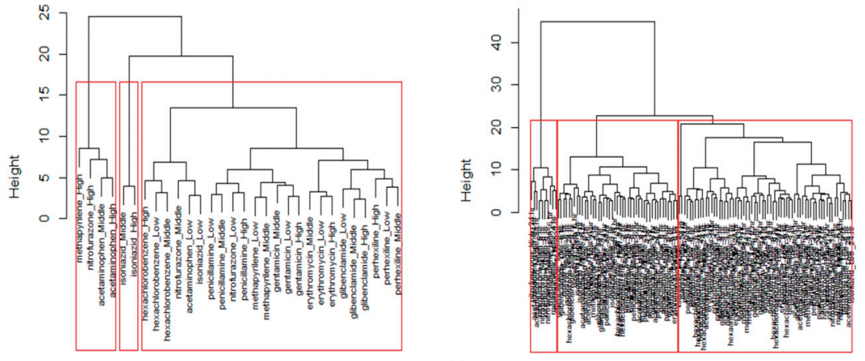
Service Process

Biomedical-Bioinformatics, as a division of CD Genomics, provides researchers with high-quality personalized data analysis and chart production services. For preclinical research analysis, CD Genomics provides bioinformatics methods such as artificial intelligence methods or statistical methods for data analysis. If you have any questions about the data analysis cycle, analysis content and price, please click online inquiry.
What's More
As one of the experienced biological information analysis service providers, CD Genomics provides established, cost-efficient, and rapid turnaround rare diseases related data analysis services for biomedical researchers. For preclinical research, CD Genomics not only provides bioinformatics data analysis services, but can also help researchers formulate appropriate technical routes and provide related technical services according to the research purposes of the researchers. For more detailed information about data analysis, please feel free to contact us. We have a professional technical support team to answer your questions.
References
- Hasan MN, et al. Assessment of Drugs Toxicity and Associated Biomarker Genes Using Hierarchical Clustering[J]. Medicina (Kaunas). 2019 Aug 8;55(8):451.
- Kaye JB, et al. Warfarin Pharmacogenomics in Diverse Populations[J]. Pharmacotherapy. 2017 Sep;37(9):1150-1163.
* For research use only. Not for use in clinical diagnosis or treatment of humans or animals.
Online Inquiry
Please submit a detailed description of your project. Our industry-leading scientists will review the information provided as soon as possible. You can also send emails directly to for inquiries.
